
Confirming The Link Between Autism Symptoms, Gut Microbes and The Brain
Autism Spectrum Disorder (ASD) is a complex condition that affects social interactions, emotional responses, sensory processing and is associated with repetitive behaviors.
The most recent data (2022) from the Center for Disease Control indicate that approximately 1 in 31 (3.2%) 8-year-old children have been diagnosed with ASD, marking a notable increase from previous years. This upward trend has been consistent over the past two decades and has been attributed to environmental factors, heightened awareness and broader diagnostic criteria.
“…scientists have long suspected that disruptions in the gut microbiome may contribute significantly to ASD symptoms.”
While the pathophysiology of ASD remains unknown, scientists have long suspected that disruptions in the gut microbiome may contribute significantly to the symptoms seen in autism.
Even though previous research has shown differences in gut bacteria and their byproducts (called metabolites) in individuals with ASD, it’s not clear exactly how these changes might influence brain activity and behaviors associated with ASD.
Our recent publication in Nature Communications from a team of investigators at USC and UCLA under the leadership of Dr. Lisa Aziz-Zadeh reports results from a study involving 84 children and teenagers, half of whom had autism and half who did not (referred to as “neurotypical,” NT). Participants ranged from 8 to 17 years old. The study collected stool samples to analyze gut metabolites, conducted MRI brain scans and performed extensive behavioral assessments.
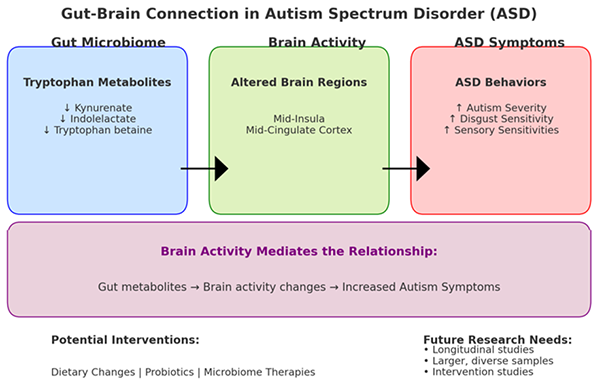
The research team was particularly interested in metabolites derived from tryptophan, an amino acid crucial for many brain and body functions. Tryptophan can be converted by gut bacteria into various chemicals. These substances are significant because some (like serotonin and kynurenine) can affect brain function and mood, while others, like kynurenate, have protective effects on the brain. This makes them strong candidates for influencing ASD symptoms.
Here are the key findings of the study:
Differences in Gut Metabolites between ASD and NT groups:
The researchers found notable differences in specific tryptophan metabolites between the autistic participants and the neurotypical group. Most significantly, kynurenate levels were lower in the stool samples of the ASD group suggesting reduced protection from brain inflammation or damage.
Links Between Metabolites and Brain Activity:
Using fMRI scans, the researchers studied brain activity while participants watched videos designed to provoke emotional and sensory reactions. These videos included social interactions, faces expressing different emotions and scenes intended to trigger feelings of disgust or discomfort. The ASD group showed distinct patterns of brain activation, especially in regions responsible for processing emotions, social cues and internal bodily sensations.
Interestingly, the altered brain activity in children with ASD was significantly related to the amounts of specific gut metabolites. For instance, lower levels of the metabolite indolelactate, another tryptophan metabolite were linked to unusual activity in a brain region called the mid-insula, which is crucial for interpreting bodily sensations and emotional states. This connection implies that changes in gut metabolites could directly or indirectly influence brain function.
Connections Between Brain Activity and Autism Symptoms:
Brain regions showing atypical activity in ASD participants were strongly linked to the severity of autism symptoms. For example, abnormal activation in the mid-insula correlated with stronger repetitive behaviors and more severe overall autism symptoms, indicating that disrupted gut metabolites might exacerbate specific ASD-related difficulties.
In addition, another brain region, the mid-cingulate cortex, played an essential role in mediating emotional sensitivity (particularly sensitivity to disgust) in autistic participants, again influenced by tryptophan-related metabolites.
Brain Activity Mediates the Relationship Between Gut Metabolites and Autism Symptoms:
A crucial part of this study was to test the hypothesis that brain activity could explain the link between gut metabolites and behavior in autism. Supporting this hypothesis, brain regions such as the mid-insula and mid-cingulate cortex seemed to mediate or connect changes in gut metabolite levels to autism symptoms. This means that gut metabolites might influence behavior indirectly by first altering brain activity.
These findings represent a significant advancement in understanding ASD pathophysiology suggesting that certain gut metabolites derived from dietary tryptophan might impact symptoms by affecting brain function.
The authors acknowledge several limitations. The study was cross-sectional, meaning it captured a snapshot in time without proving cause and effect. Moreover, the sample was relatively small and the autism group was more male-dominated, reflecting general autism demographics but potentially limiting generalizability. Additionally, although diet was measured and analyzed, dietary reporting might have inaccuracies, potentially influencing metabolite results.
“…the brain-gut-microbiome system …is crucial in mental health and neurological conditions.”
This new evidence aligns with prior research showing the brain-gut-microbiome system—the complex communication network between the gut microbiome and the brain—is crucial in mental health and neurological conditions. Gut metabolites derived from tryptophan can cross into the brain and directly influence neural pathways and brain inflammation, potentially affecting development and behavior, particularly during critical growth periods in childhood.
However, more research is needed to fully confirm these findings. Future studies should include larger, more diverse groups of participants and longitudinal designs that track changes in gut metabolites, brain activity and autism symptoms over time. Such studies could identify whether manipulating the gut microbiome through dietary interventions, probiotics, or fecal microbiota transplants could effectively improve autism symptoms.

Emeran Mayer, MD Is a Distinguished Research Professor in the Departments of Medicine, Physiology and Psychiatry at the David Geffen School of Medicine at UCLA, the Executive Director of the G. Oppenheimer Center for Neurobiology of Stress and Resilience and the Founding Director of the Goodman-Luskin Microbiome Center at UCLA.